You are here

According to the European Chemicals Agency’s (ECHA) 25 September 2019 newsletter, the Competent Authorities for REACH and CLP (CARACAL) have unanimously agreed to the European Commission’s proposed changes to Annex VIII to the Regulation on Classification, Labeling and Packaging (1272/2008/EC – CLP), including the postponement by one year of the implementation of the Poison Centre Notification (PCN) requirements for mixtures placed on the market for consumer use.
The draft agenda of CARACAL’s 31st meeting, which was scheduled for 18 September 2019, included discussions on the proposed Annex VIII changes, as well as the CLP’s upcoming 14th and 15th adaptations to technical and scientific progress (ATPs).
CARACAL is an expert group which advises the Commission and ECHA on questions related to REACH (1907/2006/EC) and CLP Regulations. The group is composed of representatives of EU member state competent authorities for REACH and CLP, representatives from competent authorities of EEA-EFTA countries, as well as several observers from non-EU countries, international organizations and stakeholders.
The Commission is now proceeding with adopting a delegated act which will, among other things, postpone the first compliance date for harmonized reporting to poison centres, for mixtures intended for consumer use, from 1 January 2020 to 1 January 2021. The other compliance deadlines concerning professional and industrial uses will not be affected. Entry into force of these changes is expected later in 2019.
ECHA adds that the Commission is also progressing with solving some of the concerns raised by stakeholders on the workability of the notification requirements. In this context, another amendment to Annex VIII is expected in 2020.
Evolving PCN Requirements
Currently, poison centre information requirements vary by country and companies are required to make separate notifications for each country. This is difficult and time consuming for businesses supplying products across Europe. The current arrangements are also not very effective. ChemicalWatch reports that in up to 40% of calls poison centres were unable to identify the mixture risking health or had difficulty doing so, causing unnecessary hospitalizations.
To resolve this Annex VIII to the CLP Regulationsets out a unified format for submitting PCN notifications to appointed bodies in EU Member States. It also adds several new and more stringent requirements for companies, increasing the burden of compliance.
According to Annex VIII importers and downstream users placing hazardous mixtures on the market must:
- Prepare and assign a 16-character Unique Formula Identifier (UFI) for each mixture formulation
- Notify all mixtures placed on the market that are classified for human health or physical hazards
- Include the UFI on the product label and/or Safety Data Sheet (SDS)
- Submit a substantial amount of product information electronically either via the ECHA PCN Portal or directly to the Member States’ appointed bodies
Streamline and Automate PCN Processes
At Verisk 3E we are working closely with ECHA to stay ahead of the upcoming changes and provide targeted solutions to streamline compliance with both current as well as new harmonized PCN requirements. We offer a suite of solutions that enables clients to improve product safety and remain in compliance with the European PCN requirements.
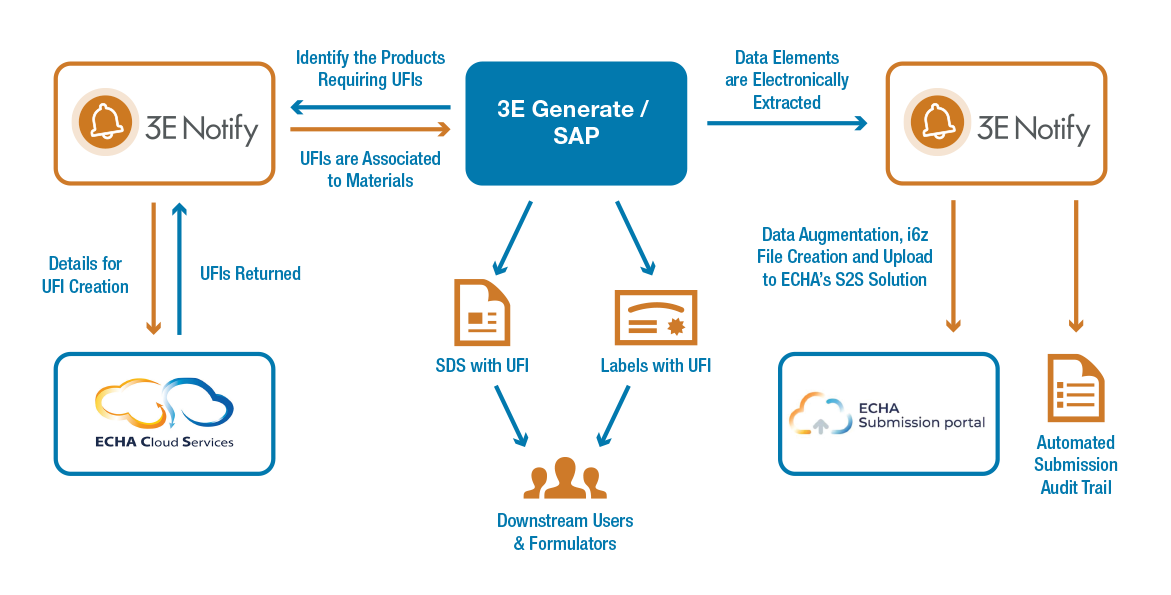
The new cloud-based 3E Notify for Poison Centres application automates time-consuming manual poison centre notification processes including UFI generation, data extraction and mapping, dossier compilation and notification submission.
3E Notify seamlessly integrates with clients’ product safety and hazard communication platforms, including 3E Generate™, Verisk 3E’s Safety Data Sheet (SDS) and label authoring system, and the product safety capabilities of SAP® Environment, Health, and Safety (EHS) Management and SAP S/4HANA® for product compliance.
In addition, Verisk 3E provides PCN services that include guidance in understanding your requirements and business impacts, planning and analysis, data obtainment, UFI strategy development, SDS and label updates and notification submission. Notification services are offered via 3E SDS Authoring, 3E Regulatory Consulting, and 3E ERD™, a cloud-based regulatory documentation solution.
Reach out to our PCN team for help preparing and complying with your PCN obligations before the deadline is upon you.

 Top
Top